(ORDO NEWS) — Scientists from the Institute of Terrestrial Microbiology of the Max Planck Society and the University of Marburg (Germany) have uncovered the evolution of the most common enzyme on Earth – rubisco, which plays a key role in the process of photosynthesis.
Rubisco or ribulose bisphosphate carboxylase (RuBisCO) catalyzes the addition of absorbed carbon dioxide to ribulose-1,5-bisphosphate, the molecule that is the starting point of the Calvin cycle, during which carbon from CO2 is taken up by plants and cyanobacteria and incorporated into their biomass.
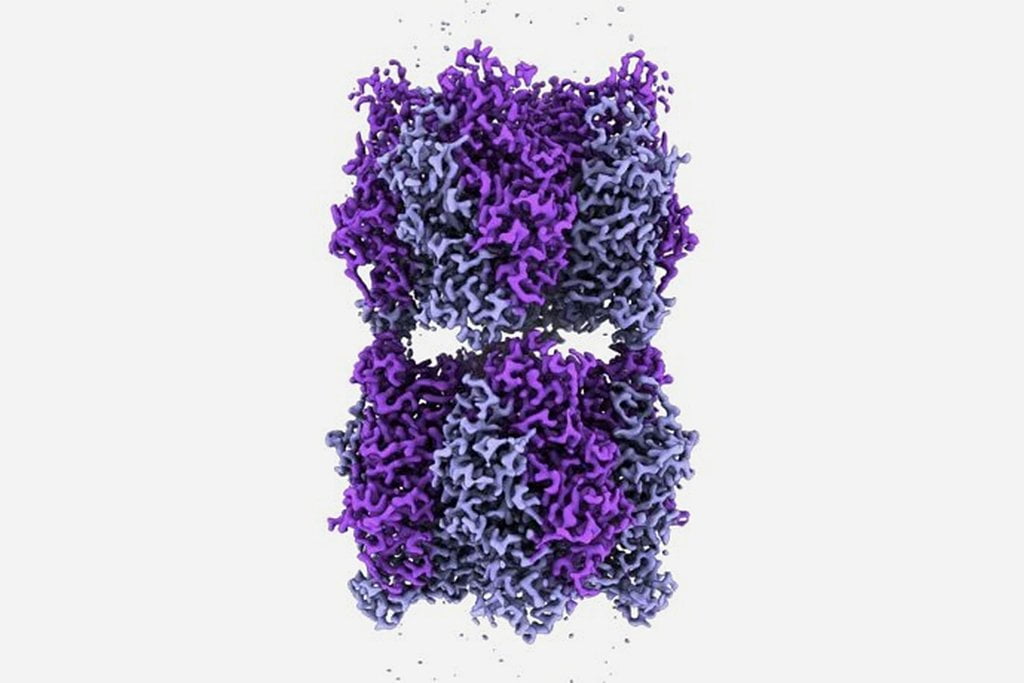
Rubisco consists of two major components, a large L subunit and a small S subunit. L has catalytic sites directly involved in carbon fixation, but the role of the S subunit has not been understood.
The researchers synthesized likely ancestral forms of rubisco that existed about a billion years ago to reconstruct an important evolutionary event when the enzyme learned to distinguish carbon dioxide from an alternative substrate, molecular oxygen.
In the past, when the Earth’s atmosphere began to become saturated with oxygen, rubisco began to catalyze an undesirable reaction, during which the assimilation of molecular oxygen took place and radicals toxic to living cells were produced.
Until now, under certain conditions, the enzyme switches to photorespiration – oxygenase activity, which reduces the efficiency of photosynthesis.
Over the course of evolution, rubisco had to become more specific to carbon dioxide in order to ensure the survival of the ancestors of modern plants and other photosynthetic organisms.
The researchers expected that ancestral forms of the enzyme acquired a new component that directly excluded oxygen from the catalytic sites of the L subunit. Instead, they found that the addition of a small subunit affects the effect that subsequent mutations of the enzyme have on the catalytic sites
Those mutations that were insignificant for the L subunit, in the presence of the S subunit, began to have a huge impact on the enzyme.
Analysis of enzymatic activity and macromolecular structures showed that the small subunit quickly became indispensable and contributed to broader functional changes in the large subunit, including increased activity and specificity for carbon dioxide.
According to the scientists, the results show that rubisco can be modified by adding new protein components and even reduce carbon loss during photorespiration.
—
Online:
Contact us: [email protected]
Our Standards, Terms of Use: Standard Terms And Conditions.